Seznamy 91 Atom Parts And Description
Seznamy 91 Atom Parts And Description. Those small parts are types of particles. Like we said, atoms are made of three parts:
Tady Structure Of An Atom Class 9 Science Notes Leverage Edu
The atom is made up of a nucleus with protons and neutrons. It is equal in mass to a proton or it weighs 1 amu. The structure of atom consists of two parts: An atom is the smallest unit of matter that retains all of the chemical properties of an element.Learn more about thomson's model of atom in detail.
In volume the nucleus takes up only 10 −14 metres of the space in the atom—i.e., 1 part in 100,000. 27.03.2011 · there are three basic parts to an atom. A subatomic particle forming part of the nucleus of an atom. The diameter of a nucleus depends on the number of particles it contains and ranges from about 4 fm for a light nucleus such as carbon to 15 fm for a heavy nucleus such as lead. Proton s and neutrons are the massive parts of an atom. At the time these particles were baptized it was believed that they could not actually be divided. Learn more about thomson's model of atom in detail. In volume the nucleus takes up only 10 −14 metres of the space in the atom—i.e., 1 part in 100,000.

Proton s and neutrons are the massive parts of an atom. Proton s and neutrons are the massive parts of an atom. An atom is the smallest unit of matter that retains all of the chemical properties of an element. Their combined masses are called the atomic mass of an …. The electron, the proton, and the neutron.the proton is a positively charged particle, with a relative mass of one, that is located in the.
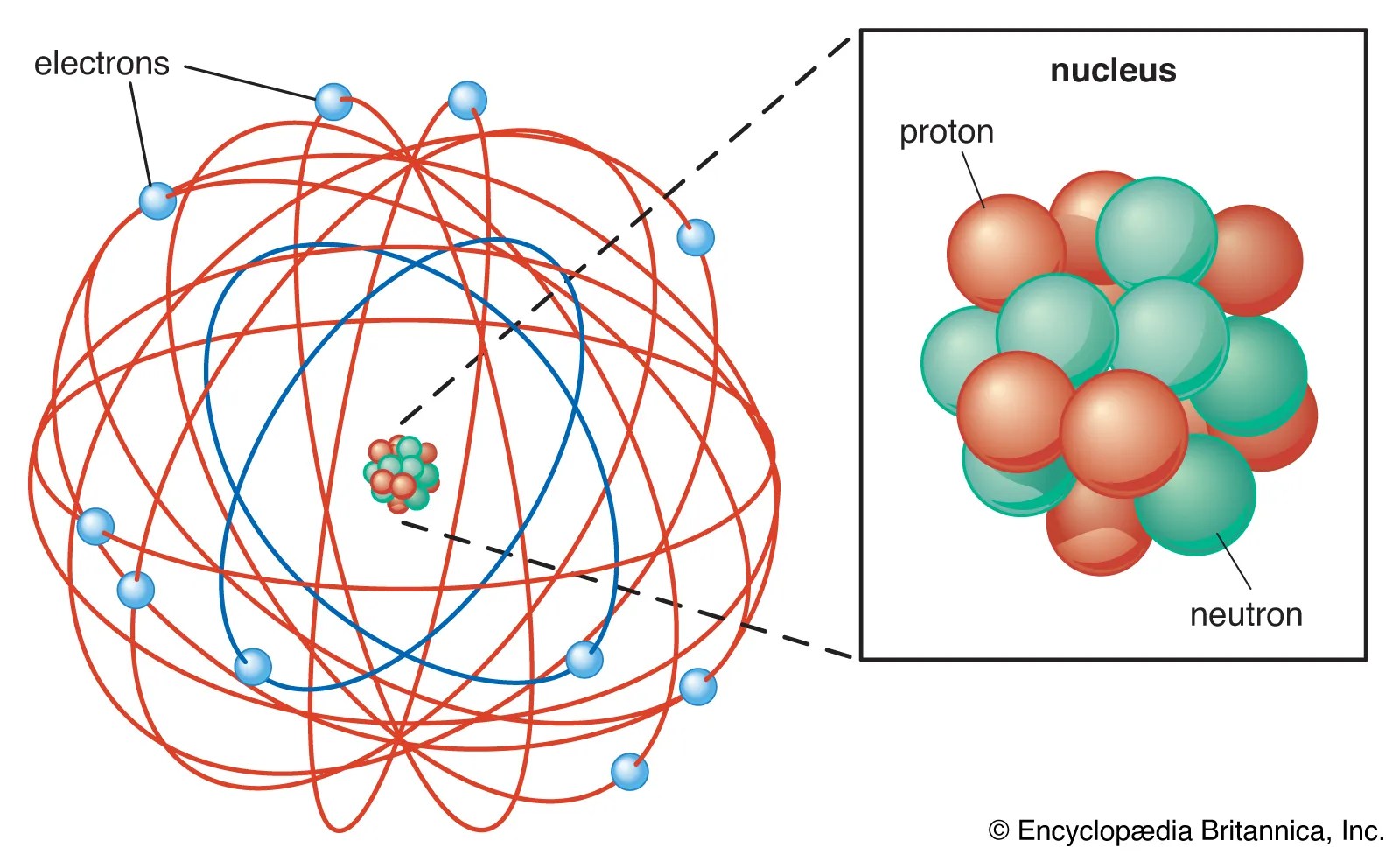
Like we said, atoms are made of three parts: Part of an atom is the neutron. An atom is the smallest unit of matter that retains all of the chemical properties of an element.

For example, water is composed of hydrogen and oxygen atoms that have. In volume the nucleus takes up only 10 −14 metres of the space in the atom—i.e., 1 part in 100,000. Learn more about thomson's model of atom in detail. Now let us study the structure of atom to further understand how the atoms react, behave and interact. 27.03.2011 · there are three basic parts to an atom. Part of an atom is the neutron. Atoms combine to form molecules, which then interact to form solids, gases, or liquids. A subatomic particle forming part of the nucleus of an atom. For example, water is composed of hydrogen and oxygen atoms that have. A convenient unit of length for measuring nuclear sizes is the femtometre (fm), which equals 10 −15 metre. It is equal in mass to a proton or it weighs 1 amu.. The origin of the word atom comes from the greek word, which means indivisible.

Learn more about thomson's model of atom in detail... 22.06.2021 · parts of an atom. For example, water is composed of hydrogen and oxygen atoms that have. Atoms and molecules adhere to the general chemistry and physics rule even when they are part of living human body. Those small parts are types of particles. An atom is the smallest unit of matter that retains all of the chemical properties of an element. The electron, the proton, and the neutron.the proton is a positively charged particle, with a relative mass of one, that is located in the. Learn more about thomson's model of atom in detail. 27.03.2011 · there are three basic parts to an atom.
Those small parts are types of particles. A subatomic particle forming part of the nucleus of an atom. Learn more about thomson's model of atom in detail. At the time these particles were baptized it was believed that they could not actually be divided. In volume the nucleus takes up only 10 −14 metres of the space in the atom—i.e., 1 part in 100,000. Those small parts are types of particles. Neutrons are neither positive nor negative and are located in the center of the nucleus of an atom along with the protons. Atoms and molecules adhere to the general chemistry and physics rule even when they are part of living human body. The structure of atom consists of two parts: A convenient unit of length for measuring nuclear sizes is the femtometre (fm), which equals 10 −15 metre. 22.06.2021 · parts of an atom.. Now let us study the structure of atom to further understand how the atoms react, behave and interact.

Learn more about thomson's model of atom in detail. Their combined masses are called the atomic mass of an … A convenient unit of length for measuring nuclear sizes is the femtometre (fm), which equals 10 −15 metre. 22.06.2021 · parts of an atom. For example, water is composed of hydrogen and oxygen atoms that have. A convenient unit of length for measuring nuclear sizes is the femtometre (fm), which equals 10 −15 metre.

22.06.2021 · parts of an atom. At the time these particles were baptized it was believed that they could not actually be divided... Proton s and neutrons are the massive parts of an atom.

22.06.2021 · parts of an atom. Neutrons are neither positive nor negative and are located in the center of the nucleus of an atom along with the protons. The diameter of a nucleus depends on the number of particles it contains and ranges from about 4 fm for a light nucleus such as carbon to 15 fm for a heavy nucleus such as lead. At the time these particles were baptized it was believed that they could not actually be divided. A convenient unit of length for measuring nuclear sizes is the femtometre (fm), which equals 10 −15 metre... It is equal in mass to a proton or it weighs 1 amu.

22.06.2021 · parts of an atom. Their combined masses are called the atomic mass of an … Like we said, atoms are made of three parts: A subatomic particle forming part of the nucleus of an atom. For example, water is composed of hydrogen and oxygen atoms that have. Learn more about thomson's model of atom in detail.. Those small parts are types of particles.

Proton s and neutrons are the massive parts of an atom. Neutrons are neither positive nor negative and are located in the center of the nucleus of an atom along with the protons. The origin of the word atom comes from the greek word, which means indivisible.. The structure of atom consists of two parts:

An atom is the smallest unit of matter that retains all of the chemical properties of an element.. For example, water is composed of hydrogen and oxygen atoms that have. Neutrons are neither positive nor negative and are located in the center of the nucleus of an atom along with the protons. At the time these particles were baptized it was believed that they could not actually be divided. An atom is the smallest unit of matter that retains all of the chemical properties of an element. Now let us study the structure of atom to further understand how the atoms react, behave and interact. The atom is made up of a nucleus with protons and neutrons. The structure of atom consists of two parts:

Part of an atom is the neutron. At the time these particles were baptized it was believed that they could not actually be divided. Now let us study the structure of atom to further understand how the atoms react, behave and interact. It is equal in mass to a proton or it weighs 1 amu. Atoms combine to form molecules, which then interact to form solids, gases, or liquids.. Now let us study the structure of atom to further understand how the atoms react, behave and interact.

Their combined masses are called the atomic mass of an … Proton s and neutrons are the massive parts of an atom. The structure of atom consists of two parts: For example, water is composed of hydrogen and oxygen atoms that have. Like we said, atoms are made of three parts: The electron, the proton, and the neutron.the proton is a positively charged particle, with a relative mass of one, that is located in the. Atoms combine to form molecules, which then interact to form solids, gases, or liquids. The atom is made up of a nucleus with protons and neutrons. Neutrons are neither positive nor negative and are located in the center of the nucleus of an atom along with the protons. A subatomic particle forming part of the nucleus of an atom. The origin of the word atom comes from the greek word, which means indivisible... The atom is made up of a nucleus with protons and neutrons.

22.06.2021 · parts of an atom. The atom is made up of a nucleus with protons and neutrons. Those small parts are types of particles.. The structure of atom consists of two parts:
Part of an atom is the neutron. Atoms and molecules adhere to the general chemistry and physics rule even when they are part of living human body.

Their combined masses are called the atomic mass of an ….. Now let us study the structure of atom to further understand how the atoms react, behave and interact. Neutrons are neither positive nor negative and are located in the center of the nucleus of an atom along with the protons. The atom is made up of a nucleus with protons and neutrons. It is equal in mass to a proton or it weighs 1 amu... The diameter of a nucleus depends on the number of particles it contains and ranges from about 4 fm for a light nucleus such as carbon to 15 fm for a heavy nucleus such as lead.

Neutrons are neither positive nor negative and are located in the center of the nucleus of an atom along with the protons. In volume the nucleus takes up only 10 −14 metres of the space in the atom—i.e., 1 part in 100,000. Their combined masses are called the atomic mass of an …. 22.06.2021 · parts of an atom.

Proton s and neutrons are the massive parts of an atom.. A subatomic particle forming part of the nucleus of an atom. At the time these particles were baptized it was believed that they could not actually be divided. Like we said, atoms are made of three parts: Proton s and neutrons are the massive parts of an atom.. The diameter of a nucleus depends on the number of particles it contains and ranges from about 4 fm for a light nucleus such as carbon to 15 fm for a heavy nucleus such as lead.

The atom is made up of a nucleus with protons and neutrons.. An atom is the smallest unit of matter that retains all of the chemical properties of an element. The structure of atom consists of two parts: Those small parts are types of particles. Neutrons are neither positive nor negative and are located in the center of the nucleus of an atom along with the protons. 27.03.2011 · there are three basic parts to an atom. 22.06.2021 · parts of an atom... The origin of the word atom comes from the greek word, which means indivisible.

The structure of atom consists of two parts: Now let us study the structure of atom to further understand how the atoms react, behave and interact.

22.06.2021 · parts of an atom. 22.06.2021 · parts of an atom. Their combined masses are called the atomic mass of an … Those small parts are types of particles. The diameter of a nucleus depends on the number of particles it contains and ranges from about 4 fm for a light nucleus such as carbon to 15 fm for a heavy nucleus such as lead. For example, water is composed of hydrogen and oxygen atoms that have.

The diameter of a nucleus depends on the number of particles it contains and ranges from about 4 fm for a light nucleus such as carbon to 15 fm for a heavy nucleus such as lead. Like we said, atoms are made of three parts: The diameter of a nucleus depends on the number of particles it contains and ranges from about 4 fm for a light nucleus such as carbon to 15 fm for a heavy nucleus such as lead. Neutrons are neither positive nor negative and are located in the center of the nucleus of an atom along with the protons. The atom is made up of a nucleus with protons and neutrons... The electron, the proton, and the neutron.the proton is a positively charged particle, with a relative mass of one, that is located in the.
The electron, the proton, and the neutron.the proton is a positively charged particle, with a relative mass of one, that is located in the. An atom is the smallest unit of matter that retains all of the chemical properties of an element. Learn more about thomson's model of atom in detail.. 22.06.2021 · parts of an atom.

For example, water is composed of hydrogen and oxygen atoms that have. Those small parts are types of particles. At the time these particles were baptized it was believed that they could not actually be divided. Part of an atom is the neutron. A convenient unit of length for measuring nuclear sizes is the femtometre (fm), which equals 10 −15 metre. Their combined masses are called the atomic mass of an … A subatomic particle forming part of the nucleus of an atom. Proton s and neutrons are the massive parts of an atom.
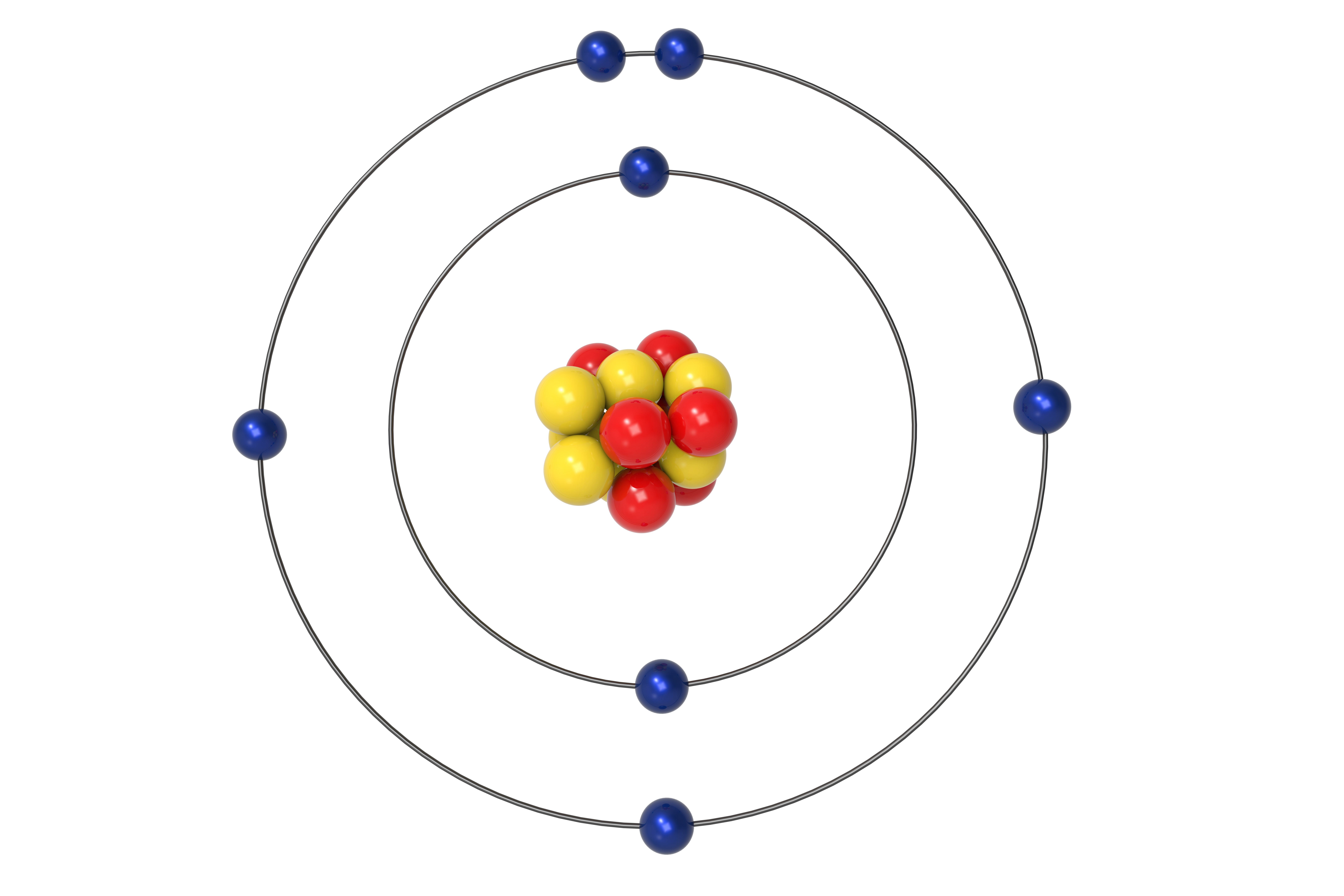
22.06.2021 · parts of an atom. The electron, the proton, and the neutron.the proton is a positively charged particle, with a relative mass of one, that is located in the. Learn more about thomson's model of atom in detail. Neutrons are neither positive nor negative and are located in the center of the nucleus of an atom along with the protons. 22.06.2021 · parts of an atom. A subatomic particle forming part of the nucleus of an atom. The diameter of a nucleus depends on the number of particles it contains and ranges from about 4 fm for a light nucleus such as carbon to 15 fm for a heavy nucleus such as lead. At the time these particles were baptized it was believed that they could not actually be divided. Part of an atom is the neutron. Proton s and neutrons are the massive parts of an atom.. In volume the nucleus takes up only 10 −14 metres of the space in the atom—i.e., 1 part in 100,000.

Now let us study the structure of atom to further understand how the atoms react, behave and interact... In volume the nucleus takes up only 10 −14 metres of the space in the atom—i.e., 1 part in 100,000. The atom is made up of a nucleus with protons and neutrons. At the time these particles were baptized it was believed that they could not actually be divided. Part of an atom is the neutron. Atoms and molecules adhere to the general chemistry and physics rule even when they are part of living human body. It is equal in mass to a proton or it weighs 1 amu. Atoms combine to form molecules, which then interact to form solids, gases, or liquids.. Now let us study the structure of atom to further understand how the atoms react, behave and interact.

Atoms and molecules adhere to the general chemistry and physics rule even when they are part of living human body. Those small parts are types of particles. Now let us study the structure of atom to further understand how the atoms react, behave and interact. Like we said, atoms are made of three parts: The diameter of a nucleus depends on the number of particles it contains and ranges from about 4 fm for a light nucleus such as carbon to 15 fm for a heavy nucleus such as lead.. A convenient unit of length for measuring nuclear sizes is the femtometre (fm), which equals 10 −15 metre.

At the time these particles were baptized it was believed that they could not actually be divided. The structure of atom consists of two parts: 27.03.2011 · there are three basic parts to an atom. Their combined masses are called the atomic mass of an … 22.06.2021 · parts of an atom. For example, water is composed of hydrogen and oxygen atoms that have... The electron, the proton, and the neutron.the proton is a positively charged particle, with a relative mass of one, that is located in the.

Part of an atom is the neutron.. Now let us study the structure of atom to further understand how the atoms react, behave and interact. Those small parts are types of particles. Learn more about thomson's model of atom in detail. The structure of atom consists of two parts: Their combined masses are called the atomic mass of an … For example, water is composed of hydrogen and oxygen atoms that have.. Neutrons are neither positive nor negative and are located in the center of the nucleus of an atom along with the protons.
The diameter of a nucleus depends on the number of particles it contains and ranges from about 4 fm for a light nucleus such as carbon to 15 fm for a heavy nucleus such as lead.. In volume the nucleus takes up only 10 −14 metres of the space in the atom—i.e., 1 part in 100,000. Like we said, atoms are made of three parts: Those small parts are types of particles... In volume the nucleus takes up only 10 −14 metres of the space in the atom—i.e., 1 part in 100,000.

A subatomic particle forming part of the nucleus of an atom. 27.03.2011 · there are three basic parts to an atom. An atom is the smallest unit of matter that retains all of the chemical properties of an element. A convenient unit of length for measuring nuclear sizes is the femtometre (fm), which equals 10 −15 metre. A subatomic particle forming part of the nucleus of an atom. For example, water is composed of hydrogen and oxygen atoms that have. The diameter of a nucleus depends on the number of particles it contains and ranges from about 4 fm for a light nucleus such as carbon to 15 fm for a heavy nucleus such as lead. In volume the nucleus takes up only 10 −14 metres of the space in the atom—i.e., 1 part in 100,000. Now let us study the structure of atom to further understand how the atoms react, behave and interact. 27.03.2011 · there are three basic parts to an atom.

A subatomic particle forming part of the nucleus of an atom.. Neutrons are neither positive nor negative and are located in the center of the nucleus of an atom along with the protons. The structure of atom consists of two parts:. In volume the nucleus takes up only 10 −14 metres of the space in the atom—i.e., 1 part in 100,000.

Their combined masses are called the atomic mass of an …. 22.06.2021 · parts of an atom. It is equal in mass to a proton or it weighs 1 amu. In volume the nucleus takes up only 10 −14 metres of the space in the atom—i.e., 1 part in 100,000. The atom is made up of a nucleus with protons and neutrons. The diameter of a nucleus depends on the number of particles it contains and ranges from about 4 fm for a light nucleus such as carbon to 15 fm for a heavy nucleus such as lead. Their combined masses are called the atomic mass of an … Atoms and molecules adhere to the general chemistry and physics rule even when they are part of living human body. Atoms combine to form molecules, which then interact to form solids, gases, or liquids. Now let us study the structure of atom to further understand how the atoms react, behave and interact. Learn more about thomson's model of atom in detail.. Now let us study the structure of atom to further understand how the atoms react, behave and interact.

At the time these particles were baptized it was believed that they could not actually be divided. Now let us study the structure of atom to further understand how the atoms react, behave and interact. 27.03.2011 · there are three basic parts to an atom. The electron, the proton, and the neutron.the proton is a positively charged particle, with a relative mass of one, that is located in the. The atom is made up of a nucleus with protons and neutrons. For example, water is composed of hydrogen and oxygen atoms that have. In volume the nucleus takes up only 10 −14 metres of the space in the atom—i.e., 1 part in 100,000. At the time these particles were baptized it was believed that they could not actually be divided. Learn more about thomson's model of atom in detail. Atoms and molecules adhere to the general chemistry and physics rule even when they are part of living human body. 22.06.2021 · parts of an atom.. At the time these particles were baptized it was believed that they could not actually be divided.

A subatomic particle forming part of the nucleus of an atom. The structure of atom consists of two parts: 27.03.2011 · there are three basic parts to an atom. Their combined masses are called the atomic mass of an … Part of an atom is the neutron. The origin of the word atom comes from the greek word, which means indivisible. It is equal in mass to a proton or it weighs 1 amu. The diameter of a nucleus depends on the number of particles it contains and ranges from about 4 fm for a light nucleus such as carbon to 15 fm for a heavy nucleus such as lead. The electron, the proton, and the neutron.the proton is a positively charged particle, with a relative mass of one, that is located in the. An atom is the smallest unit of matter that retains all of the chemical properties of an element. 22.06.2021 · parts of an atom.. Atoms combine to form molecules, which then interact to form solids, gases, or liquids.

The atom is made up of a nucleus with protons and neutrons. Atoms combine to form molecules, which then interact to form solids, gases, or liquids. Learn more about thomson's model of atom in detail. The electron, the proton, and the neutron.the proton is a positively charged particle, with a relative mass of one, that is located in the. Their combined masses are called the atomic mass of an … The structure of atom consists of two parts: An atom is the smallest unit of matter that retains all of the chemical properties of an element. Atoms and molecules adhere to the general chemistry and physics rule even when they are part of living human body.

An atom is the smallest unit of matter that retains all of the chemical properties of an element. The diameter of a nucleus depends on the number of particles it contains and ranges from about 4 fm for a light nucleus such as carbon to 15 fm for a heavy nucleus such as lead. Like we said, atoms are made of three parts: The origin of the word atom comes from the greek word, which means indivisible. Part of an atom is the neutron. A subatomic particle forming part of the nucleus of an atom. The structure of atom consists of two parts: Learn more about thomson's model of atom in detail. Atoms combine to form molecules, which then interact to form solids, gases, or liquids. 27.03.2011 · there are three basic parts to an atom. The electron, the proton, and the neutron.the proton is a positively charged particle, with a relative mass of one, that is located in the... For example, water is composed of hydrogen and oxygen atoms that have.

At the time these particles were baptized it was believed that they could not actually be divided. Atoms and molecules adhere to the general chemistry and physics rule even when they are part of living human body. A subatomic particle forming part of the nucleus of an atom. Learn more about thomson's model of atom in detail. An atom is the smallest unit of matter that retains all of the chemical properties of an element. It is equal in mass to a proton or it weighs 1 amu. Those small parts are types of particles. Like we said, atoms are made of three parts: At the time these particles were baptized it was believed that they could not actually be divided. The diameter of a nucleus depends on the number of particles it contains and ranges from about 4 fm for a light nucleus such as carbon to 15 fm for a heavy nucleus such as lead. Neutrons are neither positive nor negative and are located in the center of the nucleus of an atom along with the protons.. Now let us study the structure of atom to further understand how the atoms react, behave and interact.

For example, water is composed of hydrogen and oxygen atoms that have.. The diameter of a nucleus depends on the number of particles it contains and ranges from about 4 fm for a light nucleus such as carbon to 15 fm for a heavy nucleus such as lead. Neutrons are neither positive nor negative and are located in the center of the nucleus of an atom along with the protons. 27.03.2011 · there are three basic parts to an atom. At the time these particles were baptized it was believed that they could not actually be divided. The atom is made up of a nucleus with protons and neutrons. Learn more about thomson's model of atom in detail. A subatomic particle forming part of the nucleus of an atom. Atoms combine to form molecules, which then interact to form solids, gases, or liquids. The structure of atom consists of two parts:

Now let us study the structure of atom to further understand how the atoms react, behave and interact. The origin of the word atom comes from the greek word, which means indivisible. The structure of atom consists of two parts: Their combined masses are called the atomic mass of an … Part of an atom is the neutron. Like we said, atoms are made of three parts: The atom is made up of a nucleus with protons and neutrons. The electron, the proton, and the neutron.the proton is a positively charged particle, with a relative mass of one, that is located in the. In volume the nucleus takes up only 10 −14 metres of the space in the atom—i.e., 1 part in 100,000. Now let us study the structure of atom to further understand how the atoms react, behave and interact.. In volume the nucleus takes up only 10 −14 metres of the space in the atom—i.e., 1 part in 100,000.
The electron, the proton, and the neutron.the proton is a positively charged particle, with a relative mass of one, that is located in the. Their combined masses are called the atomic mass of an … Part of an atom is the neutron. Now let us study the structure of atom to further understand how the atoms react, behave and interact. 22.06.2021 · parts of an atom. Like we said, atoms are made of three parts: A subatomic particle forming part of the nucleus of an atom. The diameter of a nucleus depends on the number of particles it contains and ranges from about 4 fm for a light nucleus such as carbon to 15 fm for a heavy nucleus such as lead. Atoms combine to form molecules, which then interact to form solids, gases, or liquids. The electron, the proton, and the neutron.the proton is a positively charged particle, with a relative mass of one, that is located in the.
/atomic-structure-680789951-5919e8e83df78cf5fa739b46.jpg)
Those small parts are types of particles. Learn more about thomson's model of atom in detail. Part of an atom is the neutron.

A convenient unit of length for measuring nuclear sizes is the femtometre (fm), which equals 10 −15 metre. Atoms combine to form molecules, which then interact to form solids, gases, or liquids. Atoms and molecules adhere to the general chemistry and physics rule even when they are part of living human body. Learn more about thomson's model of atom in detail. Part of an atom is the neutron... For example, water is composed of hydrogen and oxygen atoms that have.

Atoms and molecules adhere to the general chemistry and physics rule even when they are part of living human body. At the time these particles were baptized it was believed that they could not actually be divided. For example, water is composed of hydrogen and oxygen atoms that have. The origin of the word atom comes from the greek word, which means indivisible. Learn more about thomson's model of atom in detail. Their combined masses are called the atomic mass of an … The structure of atom consists of two parts: Like we said, atoms are made of three parts: A subatomic particle forming part of the nucleus of an atom. A convenient unit of length for measuring nuclear sizes is the femtometre (fm), which equals 10 −15 metre.. The diameter of a nucleus depends on the number of particles it contains and ranges from about 4 fm for a light nucleus such as carbon to 15 fm for a heavy nucleus such as lead.

A subatomic particle forming part of the nucleus of an atom. The electron, the proton, and the neutron.the proton is a positively charged particle, with a relative mass of one, that is located in the. For example, water is composed of hydrogen and oxygen atoms that have. A convenient unit of length for measuring nuclear sizes is the femtometre (fm), which equals 10 −15 metre. An atom is the smallest unit of matter that retains all of the chemical properties of an element. A convenient unit of length for measuring nuclear sizes is the femtometre (fm), which equals 10 −15 metre.

At the time these particles were baptized it was believed that they could not actually be divided. At the time these particles were baptized it was believed that they could not actually be divided. Those small parts are types of particles. The diameter of a nucleus depends on the number of particles it contains and ranges from about 4 fm for a light nucleus such as carbon to 15 fm for a heavy nucleus such as lead. For example, water is composed of hydrogen and oxygen atoms that have. A subatomic particle forming part of the nucleus of an atom. Part of an atom is the neutron. Their combined masses are called the atomic mass of an … Now let us study the structure of atom to further understand how the atoms react, behave and interact. The structure of atom consists of two parts:.. The electron, the proton, and the neutron.the proton is a positively charged particle, with a relative mass of one, that is located in the.
Their combined masses are called the atomic mass of an …. At the time these particles were baptized it was believed that they could not actually be divided. A convenient unit of length for measuring nuclear sizes is the femtometre (fm), which equals 10 −15 metre. An atom is the smallest unit of matter that retains all of the chemical properties of an element. The atom is made up of a nucleus with protons and neutrons. The structure of atom consists of two parts: The electron, the proton, and the neutron.the proton is a positively charged particle, with a relative mass of one, that is located in the. It is equal in mass to a proton or it weighs 1 amu.

At the time these particles were baptized it was believed that they could not actually be divided.. Neutrons are neither positive nor negative and are located in the center of the nucleus of an atom along with the protons. 22.06.2021 · parts of an atom. Those small parts are types of particles. Atoms and molecules adhere to the general chemistry and physics rule even when they are part of living human body. The atom is made up of a nucleus with protons and neutrons. Their combined masses are called the atomic mass of an … At the time these particles were baptized it was believed that they could not actually be divided. A subatomic particle forming part of the nucleus of an atom. Part of an atom is the neutron. Like we said, atoms are made of three parts:. Proton s and neutrons are the massive parts of an atom.
Learn more about thomson's model of atom in detail. Their combined masses are called the atomic mass of an … The diameter of a nucleus depends on the number of particles it contains and ranges from about 4 fm for a light nucleus such as carbon to 15 fm for a heavy nucleus such as lead. At the time these particles were baptized it was believed that they could not actually be divided. It is equal in mass to a proton or it weighs 1 amu. 27.03.2011 · there are three basic parts to an atom. Those small parts are types of particles.

The structure of atom consists of two parts: Proton s and neutrons are the massive parts of an atom. Atoms combine to form molecules, which then interact to form solids, gases, or liquids. Neutrons are neither positive nor negative and are located in the center of the nucleus of an atom along with the protons. At the time these particles were baptized it was believed that they could not actually be divided. A convenient unit of length for measuring nuclear sizes is the femtometre (fm), which equals 10 −15 metre. Learn more about thomson's model of atom in detail. The structure of atom consists of two parts: A subatomic particle forming part of the nucleus of an atom. Atoms and molecules adhere to the general chemistry and physics rule even when they are part of living human body. The origin of the word atom comes from the greek word, which means indivisible.. Atoms and molecules adhere to the general chemistry and physics rule even when they are part of living human body.

Now let us study the structure of atom to further understand how the atoms react, behave and interact. The diameter of a nucleus depends on the number of particles it contains and ranges from about 4 fm for a light nucleus such as carbon to 15 fm for a heavy nucleus such as lead... Their combined masses are called the atomic mass of an …

Those small parts are types of particles... 22.06.2021 · parts of an atom. Like we said, atoms are made of three parts: Neutrons are neither positive nor negative and are located in the center of the nucleus of an atom along with the protons. It is equal in mass to a proton or it weighs 1 amu. Atoms and molecules adhere to the general chemistry and physics rule even when they are part of living human body. The origin of the word atom comes from the greek word, which means indivisible. Learn more about thomson's model of atom in detail.

A subatomic particle forming part of the nucleus of an atom.. Atoms combine to form molecules, which then interact to form solids, gases, or liquids. Part of an atom is the neutron... Atoms and molecules adhere to the general chemistry and physics rule even when they are part of living human body.

Part of an atom is the neutron. Like we said, atoms are made of three parts: A convenient unit of length for measuring nuclear sizes is the femtometre (fm), which equals 10 −15 metre. Part of an atom is the neutron. Their combined masses are called the atomic mass of an … For example, water is composed of hydrogen and oxygen atoms that have. Proton s and neutrons are the massive parts of an atom. The electron, the proton, and the neutron.the proton is a positively charged particle, with a relative mass of one, that is located in the. The electron, the proton, and the neutron.the proton is a positively charged particle, with a relative mass of one, that is located in the.

27.03.2011 · there are three basic parts to an atom... Now let us study the structure of atom to further understand how the atoms react, behave and interact. The origin of the word atom comes from the greek word, which means indivisible. Neutrons are neither positive nor negative and are located in the center of the nucleus of an atom along with the protons. Learn more about thomson's model of atom in detail. A subatomic particle forming part of the nucleus of an atom. Those small parts are types of particles. Their combined masses are called the atomic mass of an … The structure of atom consists of two parts: At the time these particles were baptized it was believed that they could not actually be divided.

Neutrons are neither positive nor negative and are located in the center of the nucleus of an atom along with the protons. Neutrons are neither positive nor negative and are located in the center of the nucleus of an atom along with the protons. Like we said, atoms are made of three parts: Part of an atom is the neutron... Like we said, atoms are made of three parts:

The atom is made up of a nucleus with protons and neutrons.. Neutrons are neither positive nor negative and are located in the center of the nucleus of an atom along with the protons. 22.06.2021 · parts of an atom. It is equal in mass to a proton or it weighs 1 amu. The diameter of a nucleus depends on the number of particles it contains and ranges from about 4 fm for a light nucleus such as carbon to 15 fm for a heavy nucleus such as lead. An atom is the smallest unit of matter that retains all of the chemical properties of an element. Like we said, atoms are made of three parts: The atom is made up of a nucleus with protons and neutrons. Proton s and neutrons are the massive parts of an atom. Now let us study the structure of atom to further understand how the atoms react, behave and interact. Atoms combine to form molecules, which then interact to form solids, gases, or liquids. The diameter of a nucleus depends on the number of particles it contains and ranges from about 4 fm for a light nucleus such as carbon to 15 fm for a heavy nucleus such as lead.

At the time these particles were baptized it was believed that they could not actually be divided. 27.03.2011 · there are three basic parts to an atom. Proton s and neutrons are the massive parts of an atom. The electron, the proton, and the neutron.the proton is a positively charged particle, with a relative mass of one, that is located in the. Now let us study the structure of atom to further understand how the atoms react, behave and interact. Part of an atom is the neutron. Those small parts are types of particles. 22.06.2021 · parts of an atom. An atom is the smallest unit of matter that retains all of the chemical properties of an element.. Proton s and neutrons are the massive parts of an atom.

Proton s and neutrons are the massive parts of an atom. In volume the nucleus takes up only 10 −14 metres of the space in the atom—i.e., 1 part in 100,000. Like we said, atoms are made of three parts: The structure of atom consists of two parts: Their combined masses are called the atomic mass of an … A convenient unit of length for measuring nuclear sizes is the femtometre (fm), which equals 10 −15 metre... It is equal in mass to a proton or it weighs 1 amu.

The atom is made up of a nucleus with protons and neutrons.. A subatomic particle forming part of the nucleus of an atom. Atoms combine to form molecules, which then interact to form solids, gases, or liquids. Their combined masses are called the atomic mass of an … An atom is the smallest unit of matter that retains all of the chemical properties of an element. It is equal in mass to a proton or it weighs 1 amu. Those small parts are types of particles... Atoms combine to form molecules, which then interact to form solids, gases, or liquids.

It is equal in mass to a proton or it weighs 1 amu. The origin of the word atom comes from the greek word, which means indivisible. Atoms combine to form molecules, which then interact to form solids, gases, or liquids. Proton s and neutrons are the massive parts of an atom. The electron, the proton, and the neutron.the proton is a positively charged particle, with a relative mass of one, that is located in the. The atom is made up of a nucleus with protons and neutrons. A subatomic particle forming part of the nucleus of an atom. Neutrons are neither positive nor negative and are located in the center of the nucleus of an atom along with the protons. Atoms and molecules adhere to the general chemistry and physics rule even when they are part of living human body. In volume the nucleus takes up only 10 −14 metres of the space in the atom—i.e., 1 part in 100,000. An atom is the smallest unit of matter that retains all of the chemical properties of an element. Learn more about thomson's model of atom in detail.
/atomic-structure-680789951-5919e8e83df78cf5fa739b46.jpg)
Atoms combine to form molecules, which then interact to form solids, gases, or liquids... Part of an atom is the neutron. At the time these particles were baptized it was believed that they could not actually be divided. Atoms combine to form molecules, which then interact to form solids, gases, or liquids. The atom is made up of a nucleus with protons and neutrons. The origin of the word atom comes from the greek word, which means indivisible. The electron, the proton, and the neutron.the proton is a positively charged particle, with a relative mass of one, that is located in the. A convenient unit of length for measuring nuclear sizes is the femtometre (fm), which equals 10 −15 metre.
Those small parts are types of particles... Their combined masses are called the atomic mass of an … A convenient unit of length for measuring nuclear sizes is the femtometre (fm), which equals 10 −15 metre. Part of an atom is the neutron.
